
The regular, preoperative use of antiplatelet medications like aspirin and clopidogrel have been associated with increased perioperative blood loss as well as with the need for blood products in surgery patients—as such, a thorough understanding of current guidelines pertaining to their use prior to surgery is requisite. With the advent of newer anticoagulants (e.g., direct thrombin inhibitors, direct factor Xa inhibitors), insight into what to do about these drugs must be assimilated with knowledge of older, well-established guidelines on the cessation of traditional vitamin K antagonists.
Aspirin irreversibly inhibits cyclooxygenase-1 and -2, leading to decreased formation of thromboxane A2 and ultimately to inhibited platelet aggregation. The ability of aspirin to irreversibly induce this qualitative platelet defect has led to its widespread use as a thromboprophylaxing agent in many cardiovascular disease states (eg, coronary artery disease, carotid artery stenosis, after prosthetic valve insertion, or CABG); as such, aspirin is a very frequently encountered drug in surgical patient populations.
ADENOSINE DIPHOSPHATE RECEPTOR INHIBITORS
These antiplatelet drugs (clopidogrel, prasugrel, ticagrelor, and ticlopidine) inhibit the P2Y12 subtype of platelet adenosine diphosphate (ADP) receptors, thus causing irreversible platelet inhibition (of note, ticagrelor is an allosteric antagonist, making its ADP blockage reversible). Members of this class of drugs are frequently used in combination with other antiplatelet agents like aspirin as part of dual antiplatelet therapies for acute coronary syndromes and for thromboprophylaxis in those with stents and/or cerebrovascular disease. ADP receptor inhibitors are felt to confer greater bleeding and transfusion risks than aspirin; as such, guidelines recommend discontinuation of these agents as few as 3 days prior to surgery (with specific timing determined by a given drug’s half-life of elimination).
GLYCOPROTEIN IIb/IIIa INHIBITORS
Members of this drug class (eg, abciximab, eptifibatide, tirofiban) prevent platelet aggregation via inhibition of glycoprotein (GP) IIb/IIIa receptors on the surface of platelets; these agents are frequently used during percutaneous coronary interventions and in the treatment of acute coronary syndromes. Like the other previously mentioned high-intensity antiplatelet drugs, GP IIb/IIIa inhibitors are associated with increased bleeding after operations; as such, these medications should be stopped prior to surgery in order to decrease minor and major bleeding events. Exact timing again depends on the half-life of each agent in question.
Unfractionated heparin is the notable exception to the cessation recommendations pertaining to the high-intensity antithrombotic drugs: unfractionated heparin is the only agent which can either be discontinued shortly before operation or not at all.
Vitamin K antagonists are anticoagulants that reduce hepatic production of coagulation factors II, VII, IX, and X as well as of proteins C and S—all of which depend on vitamin K for their synthesis. Warfarin, the most widely encountered of these drugs, is used both in the prophylaxis and treatment of thromboembolic disorders (eg, venous or pulmonary clots, prosthetic valve thrombosis); warfarin is similarly used in atrial fibrillation and can serve as an adjunct to reduce systemic embolic risks after myocardial infarction. Warfarin should be discontinued 2 to 4 days before surgery and patients at higher risk of thrombosis should be bridged with intravenous heparin once the international normalized ratio becomes subtherapeutic.
NOVEL ORAL ANTICOAGULANTS: DIRECT THROMBIN INHIBITORS AND DIRECT FACTOR XA INHIBITORS
Novel oral anticoagulants (NOACs) belonging to the direct thrombin inhibitor (eg, dabigatran) and direct factor Xa inhibitor (rivaroxaban, apixaban, edoxaban) classes are used commonly, as their ease of use and their lack of a monitoring requirement (as compared to warfarin) have increased the popularity of these drugs for patients requiring long-term anticoagulation. Current guidelines regarding the perioperative use of NOACs recommend these drugs be discontinued 2 to 5 days prior to procedures with a high risk of bleeding, including major abdominal, cardiovascular, and thoracic operations. Since the diminution of the anticoagulant effects of NOACs is predictable after their cessation, bridging is typically not required after NOACs are stopped.
The management of patients on NOACs requiring emergency surgery is complicated by the fact that these agents have no specific antidote. In this setting, surgery should be deferred for at least 12 hours if at all possible; given NOACs’ short half-lives, this should allow for some mitigation of bleeding risk. If delaying surgery is not possible, expert opinion suggests the use of oral activated charcoal or hemodialysis; prophylactic administration of fresh frozen plasma (FFP) or prothrombin complex concentrates is not recommended in the absence of major bleeding.
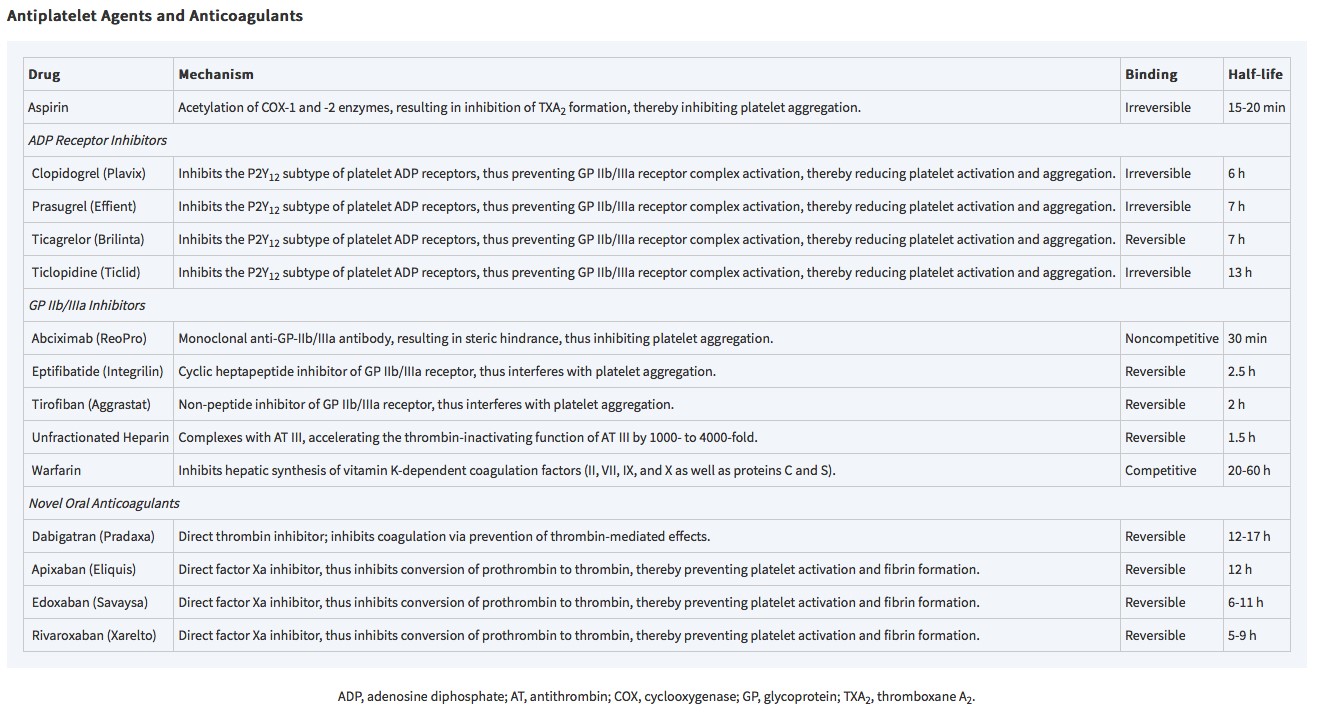
Table 14-1: Antiplatelet agents and anticoagulants (from Cohn L.H., & Adams D.H.(Eds.), (2017). Cardiac Surgery in the Adult, 5e. McGraw-Hill.)
Board Review Questions
1. What is the best laboratory test for determine degree of anticoagulation with dabigatran and rivaroxaban?
A. Prothrombin time/international normalized ratio (PT/INR)
B. Partial thromboplastin time (PTT)
C. Bleeding time
D. None of the above
2. A fully heparinized patient develops a condition requiring emergency surgery. After stopping the heparin, what else should be done to prepare the patient?
A. Nothing, if the surgery can be delayed for 2 to 3 hours.
B. Immediate administration of protamine 5 mg for every 100 units of heparin most recently administered.
C. Immediate administration of FFP.
D. Transfusion of 10 units of platelets.
3. What are the uses of thromboelastography (TEG)?
A. Predicting need for lifesaving interventions after arrival for trauma
B. Predicting 24-hour and 30-day mortality following trauma
C. Predicting early transfusion of RBC, plasma, platelets, and cryoprecipitate
D. All of the above

Create a Free MyAccess Profile
AccessMedicine Network is the place to keep up on new releases for the Access products, get short form didactic content, read up on practice impacting highlights, and watch video featuring authors of your favorite books in medicine. Create a MyAccess profile and follow our contributors to stay informed via email updates.
Excellent summary. Could we have answers for your review questions please?
2. A fully heparinized patient develops a condition requiring emergency surgery. After stopping the heparin, what else should be done to prepare the patie
Answer plz?