NAPLEX Question of the Week: Calories and Calculations!

You are a hospital pharmacist responsible for making TPN formulations at your hospital. You currently have D5W and D70W stock solutions available. D50W is typically used to compound their TPN orders, but unfortunately, it's gone on backorder so you’ll need to compound it. You need to make enough to supply the needs for the day, totaling up to 6800 kcal of D50W. How much of the D5W and D70W solutions do you need to mix to prepare the day’s supply?
A. 632 mL D5W, 2812 mL D70W
B. 912 mL D5W, 3088 mL D70W
C. 1231 mL D5W, 2769 mL D70W
D. 1394.5 mL D5W, 3138.5 mL D70W
Answer with rationale:
The correct answer is C.
Dextrose is equivalent to 3.4 kcal/ gram. D5W solutions contain 5 g / 100 mL, D50W contains 50 g / 100 mL, and D70W contains 70 g / 100 mL. The resident wants 6800 kcal of D50W, which has 6800 kcal / 3.4 kcal/g Dextrose = 2000 g Dextrose needed. As this needs to be D50W, that means the prepared solution will be 50g/100 mL and you can set up a proportion to determine the final volume of D50W: 2000 g / x mL = 50 g / 100 mL, solving for x gives you a final volume of 4000 mL. From here, the easiest way to solve this type of problem is to utilize allegation, sometimes referred to as “Tic-Tac-Toe math”.
The first step is to draw two vertical parallel lines, placing the desired concentration (i.e. D50W is 50%) in the center. From there, put the two concentrations you have on the left side, the highest concentration in the top left and the lower in the bottom left. From there, subtract the desired concentration from the top left concentration and write that on the bottom right-hand corner. Next, subtract the lower-left concentration from the desired concentration in the middle and write that number in the top right. Total up the two numbers on the right-hand side and write that below. The total volume we are trying to make in this instance is 4000 mL, so we set that equal to the total of the right side.
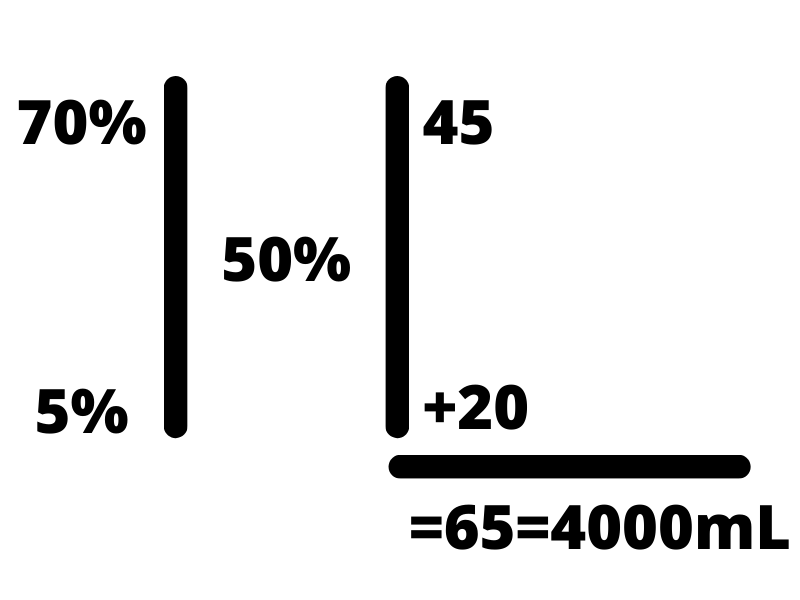
From there, we want to set up proportions to determine how much of each component we need. To do this, we draw two horizontal lines across the problem, one below the top concentration and one above the lower concentration in order to section off each component of the solution. From there, we set up a fraction with the number on the right as the numerator, and the total of the right side as the denominator, and set both fractions equal to x out of the total we desire (4000 mL in this case).
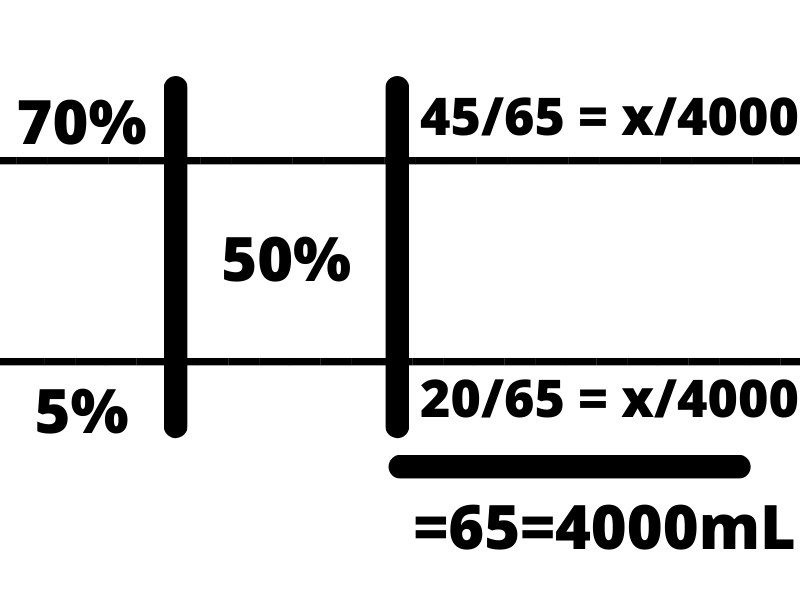
From here, it is as simple as multiplying by the desired volume and solving the problem!

Making answer choice C correct.
Answer choice A, while it adds up to 6800 kcal, the resulting solution is 2000 g / 3444 mL or D58W so it is incorrect. Answer choice B is correctly 4000 mL, but it does not add up to 6800 kcal and it results in a D55W solution. Answer choice D utilizes dextrose with an incorrect conversion of 3 kcal/gram of dextrose and results in 7700 kcal instead of 6800, making it incorrect.
Competencies covered:
4.5 – Drug concentrations, ratio strengths, osmolarity, osmolality, or extent of ionization
4.6 – Quantities of drugs or ingredients to be compounded
4.7 – Nutritional needs and the content of nutrient sources
Have a great week! Graduation is almost here!
Dr. B

Create a Free MyAccess Profile
AccessMedicine Network is the place to keep up on new releases for the Access products, get short form didactic content, read up on practice impacting highlights, and watch video featuring authors of your favorite books in medicine. Create a MyAccess profile and follow our contributors to stay informed via email updates.