Treatment of critical limb ischemia
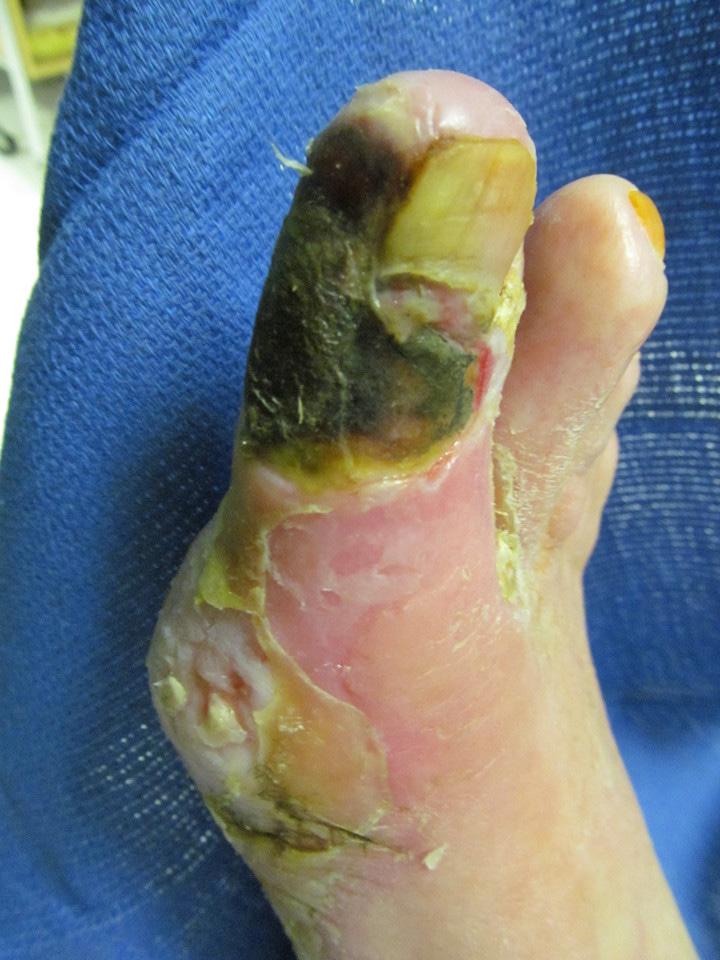
The advanced stage of peripheral arterial disease (PAD) is termed critical limb ischemia (CLI) and is characterized by rest pain (usually beginning at night when the lower extremity is extended and blood pressure tends to be lower), tissue loss or gangrene, and hypo-perfusion of the lower extremity. CLI is associated with increased risks of mortality, amputation, and impaired ADLs. Treatment of any wound associated with CLI, such as seen in the attached photo, should be conservative and protective with an immediate referral to a vascular specialist. Aggressive wound care, including debridement of non-infected necrotic tissue, is contraindicated until successful revascularization has been achieved. Debridement of a wound with hypoxic periwound tissue will only make the wound larger.
The most recent classification system for CLI, developed by the Society for Vascular Surgery Lower Extremity Guidelines Committee, is a “risk stratification based on wound, ischemia, and foot infection (WIfI)”. (Mills, 2014) Criteria for each characteristic and the numerical values are as follows:
- Wound
- 0: No ulcer and no gangrene
- 1: Small ulcer and no gangrene
- 2: Deep ulcer or gangrene limited to the toes
- 3: Extensive ulcer or extensive gangrene
- Ischemia (based on toe pressures)
- 0: >60 mmHg
- 1: 40-59mmHg
- 2: 30-39mmHg
- 3: <30 mmHg
- Foot infection
- 0: Non-infected
- 1: Mild (<2 cm cellulitis)
- 2: Moderate (>2 cm cellulitis)
- 3: Severe (systemic response/sepsis)
While the hemodynamic cut-points need more research for validity, the system does allow for risk stratification and measurable outcomes after treatment. (Shishehbor, 2016). A study by Reed et al found that an increase in ABI of 0.23 after a revascularization procedure had a significant association with increased wound healing and less need for subsequent percutaneous transluminal angioplasty. (Reed, 2017) This increased ABI value can also give direction to the wound care clinician caring for the CLI wound after revascularization.
Medical management: The medical management of the patient with CLI includes complete smoking cessation, high-dose statin, antiplatelet therapy, and antihypertensive therapy in order reduce the risk of adverse cardiovascular events. (Shishehbor, 2016)
Wound management: Debridement prior to revascularization is limited to only infected or detached necrotic tissue. The wounded area should be protected from external contaminants that may lead to infection and from shear/friction/pressure mechanical forces. Lamb’s wool or dry gauze placed between the toes helps decrease friction and maceration of interdigital skin. Collagen dressings, cell therapies, and negative pressure wound therapy (NPWT) is contraindicated at this point of care. In addition, exercise is recommended to be limited to functional activity (e.g. ambulation to the bathroom, transfer training). After revascularization, if full debridement is not performed surgically, a good indication of when to begin aggressive debridement is the presence of granulation tissue at the edge of the wound. That is a good clinical sign of sufficient perfusion to support tissue synthesis. Using vascular testing values is also recommended. Once the wound is clean and showing signs of good granulation, advanced therapies (e.g. cellular/tissue dressings, NPWT) can be initiated. Throughout all wound care, off-loading of the wounded tissue, infection and edema management (with non-elastic bandages), and continued assessment of the graft patency (using palpation or a Doppler) is essential. Any change in the graft pulse should be reported to the vascular surgeon immediately.
After the wound has healed, the patient should be fitted with an appropriate adaptive shoe or orthotic to prevent re-injury of the closed wound, keeping in mind that the wound is not fully healed until it has completed the remodeling phase discussed in Week 3. The ultimate goals of CLI interventions are to optimize function, improve quality of life, and prevent amputation.
Mills JL, Conte MS, Armstrong DA, et al. The Society for Vascular Surgery Lower Extremity Threatened Limb Classification System: risk stratification based on wound, ischemia, and foot infection (WIfI). J Vasc Surg. 2014;59:220-234.
Reed GW, Yound L, Bagh I, et al. Hemodynamic assessment before and after endovascular therapy for critical limb ischemia and association with clinical outcomes. JACC: Cardiovascular Interventions. 2017;10(23):2451-7.
Shishehbor MH, White CJ, Gray BH et al. Critical limb ischemia: an expert statement. Journal of American College of Cardiology. 2016;68(18). DOI:10.1016/j.jacc.2016.04.071.
More information is available through the following link: Woelfel S, Ochoa C, Rowe VL. Vascular wounds. In Hamm R (Ed), Text and Atlas of Wound Diagnosis and Treatment: 2nd edition. New York: McGraw Hill Education. 2019, 101-143. Available at https://accessphysiotherapy.mhmedical.com/book.aspx?bookid=1334.





Create a Free MyAccess Profile
AccessMedicine Network is the place to keep up on new releases for the Access products, get short form didactic content, read up on practice impacting highlights, and watch video featuring authors of your favorite books in medicine. Create a MyAccess profile and follow our contributors to stay informed via email updates.